Tell us what you think
We welcome your views on our website and invite you to take part in a brief survey when you've finished your visit.
Your response will help us improve the site and the experience we offer to visitors.
News and events
Find out about our free patient information events and the latest on what's happening around the hospital.
- 1
- 2
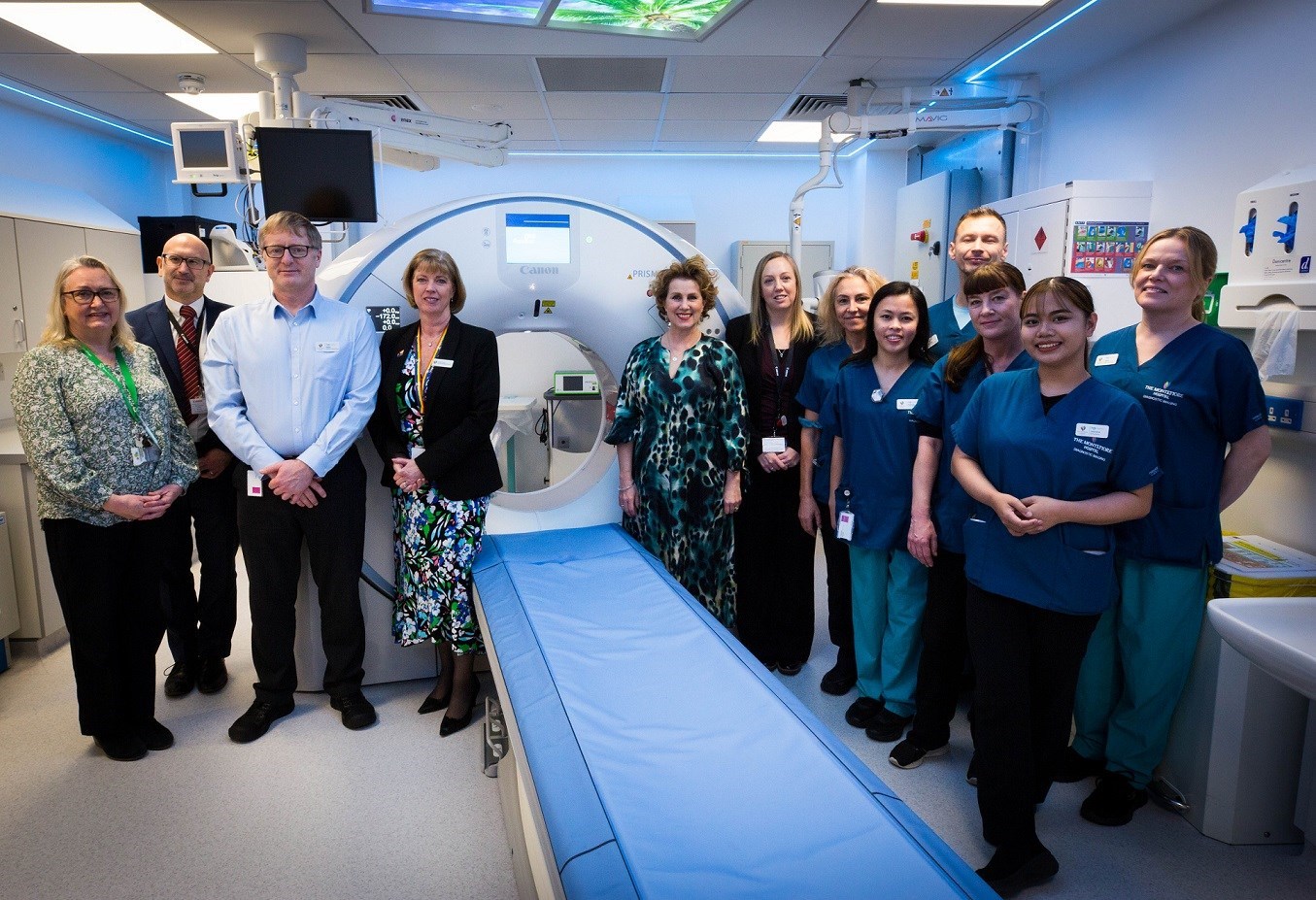
The Montefiore Hospital welcomes local author and previous patient to unveil new CT scanner
24 January 2025
The Montefiore Hospital has unveiled a new, state-of-the-art CT scanner, which will speed up diagnoses for people with underlying health concerns, such as those on a cancer pathway, or with a cardiac condition.
The Montefiore Hospital commissions bench made from 26kg of recycled plastic
14 June 2024
The Montefiore Hospital has commissioned a bench made from 26kg of its plastic waste to support Spire Healthcare’s sustainability commitment to become carbon neutral by 2030.

The Montefiore Hospital recognised for outstanding patient care from patient reviews
05 April 2024
The Montefiore Hospital has been awarded a Certificate of Excellence for outstanding patient care from an independent review site.

The Montefiore Hospital award by the National Joint Registry
14 December 2023
The Montefiore Hospital is celebrating after being named as a National Joint Registry (NJR) Gold-Level Quality Data Provider after successfully completing a national programme of local data audits.

The endoscopy department at The Montefiore announce JAG accreditation
11 September 2023
The Montefiore Hospital is excited to announce that it’s endoscopy department has achieved JAG (Joint Advisory Group) accreditation, highlighting clinical excellence for endoscopic examinations such as gastroscopy and colonoscopy.

Endoscopy team performs first case using Bravo reflux monitoring system
10 August 2022
Earlier this month, our Endoscopy team led by Consultant Gastroenterologist Dr Susi Green, performed their very first case using the Bravo™ reflux monitoring system.
Brighton resident is back on the slopes after knee replacement!
12 May 2022
Brighton resident Anthony Rogers, 60, says he was `immediately a better skier’ when he took to the slopes not long after his knee replacement.

Consultant podcasts: Dr Angus Nisbet
19 September 2019
Our Consultant Neurologist and Sleep Physician, Dr Angus Nisbet talks with The London Podcast's Adrian Lacey in this two-part discussion on consciousness.
Matt walks tall after his life-changing op
31 May 2018
It’s National Walking Month, and while most of us are happy to go on a gentle stroll, one Horsham resident takes it to the extreme…despite having had a total hip replacement.

- 1
- 2


